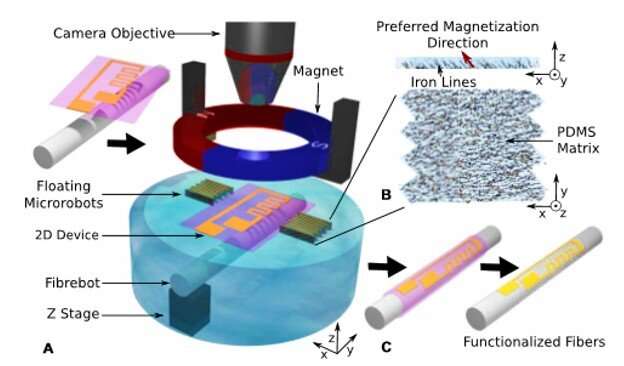
Minimally invasive surgery is increasingly used to target small lesions and a growing demand exists for miniaturized medical tools. These include microcatheters, articulated micro-forceps or tweezers to sense and actuate during precision surgery. The accurate integration and functionalization of chemical and physical sensors still remain a major challenge. In a new study on Science Robotics, Antoine Barbot and colleagues at the Institute of Medical Robotics in China and the Hamlyn Centre for Robotic Surgery in London developed a novel microrobotic platform to functionalize fibers ranging from 140 to 830 micrometers (µm). They then aligned the 2 mm x 3 mm and 200 µm-thick microbots to floating electronic circuits on a fiber using a wet transfer process.
The scientists controlled the position and orientation of the microrobots at the air-water interface using a permanent magnet. Using the nonhomogeneous magnetic field of the magnet they controlled the precise distance between the two microrobots and facilitated maneuvers of “grab and release” with floating electronic patterns. Barbot et al. proposed a model of this control process, including interactions of the microrobots through surface tension for detailed performance validation. They demonstrated a variety of example sensor embodiments on a 200 µm diameter fiber and 3-D devices.
The clinical emphasis on improved medical surveillance and diagnosis has steered the future of surgery toward precision intervention. The recent introduction of robotic tools on fibers to form fiberbots has allowed researchers to combine imaging, sensing and micromanipulation within a single fiber. Sophisticated microgrippers can be directly engineered on the tip of a fiber using two-photon polymerization. Researchers can establish microactuation using hydraulic links that leverage microcapillary function to use the device for targeted drug delivery and focused energy such as laser ablation. Optical fibers are a versatile substrate to develop flexible microtools. Their surfaces provide an ideal location to include multiple sensors along its length.
However, the direct patterning of microelectronics onto small, curved objects used for clinical applications is challenging, since existing microfabrication processes are primarily tailored to flat substrates. Researchers have hitherto used two main transfer methods; including dry transfer and the wet transfer technique. Dry transfer typically offers better cleanliness and higher precision compared to wet transfer due to the absence of wet etchant and fluid perturbation. Wet transfer techniques likely to occur at surgical interventions are limited by difficulties of accurately positioning and scooping floating devices. This is due to a lack of precision tools or robotic platforms for a precise practical approach. Microrobot manipulators can therefore address some of the major issues faced by manual wet transfer methods.
In the present work, Barbot et al. proposed the use of mobile microrobots at the air/water interface to precisely control pattern movement during wet transfer. To facilitate simple and precise control, the scientists proposed to develop a pair of microrobots to grasp floating patterns. They achieved this using a magnetic field generated with a single permanent magnet coupled to tune the magnetization direction of the robot’s constituent material. Barbot et al. used the interaction between the microrobots and the magnetic field to position the microrobot pair at two different sites of the air/water interface and form a gripper.
The researchers controlled the distance between the two microrobots by controlling the distance between the device and the magnet. They constructed the devices using an elastomer and polydomethylsiloxane (PDMS) mixed with iron powder. For a given experiment, Barbot et al. magnetized each microrobot to react similarly to a given magnetic field. The scientists used a 200 µm layer of the PDMS and iron mixture under the influence of a weak magnetic field to assemble iron lines into the PDMS matrix. They thermally cured the PDMS to keep the iron line structures in place and evaluated the orientation of the product using X-ray microtomography.
The scientists developed pairs of microrobots with custom magnetization directions using different parts of the iron/PDMS layers to form rectangular robots measuring 2mm by 3mm at 200 µm thickness. They added small tooth-like structures to the microrobots to limit the movement of the pattern during grasping processes. The team observed the positions of different microrobot pairs and their respective response to different vertical positions of the magnet to obtain two motion patterns for the microrobot pairs, depending on the angle (α) between their magnetization and the horizontal plane.
Barbot et al. designed the microrobots and allowed the height of the magnet to control the distance between the two robots and regulate opening and closing of microrobot grippers. The scientists compared the experimental results with a simulation to understand the interaction between the microrobots. They observed four degrees of freedom (DOFs) to control the microrobot pairs. These included grasping (one DOF), positioning (two DOFs) and orientations (one DOF) during wet transfer. The scientists manipulated the floating pattern using these four controllable inputs. They characterized the magnetization property of the iron/PDMS mixture as a general guide for consistent microrobot designs.
During the experiments, the team first lowered the magnet to move the microrobots apart and position them around the target pattern. They then raised the magnet to close the gap between the microrobots and grasp the pattern for subsequent alignment to the target fiber substrate. The grasping force of the microrobots did not cause any deformation of the pattern. Finally, they lowered the magnet again for the gripper to release the pattern and move the robot pair away. Using the setup, the researchers performed wet transfer of the floating pattern onto an optical fiber. During the six-step, microrobot fabrication process, the scientists transferred arbitrary gold (Au) patterns such as electrodes or strain gauges onto the fiber. To understand the precision of pattern transfer, they measured the shift between two complementary patterns deposited on the fiber. Barbot et al. observed challenges to long-term pattern adhesion on curved contact locations of the fiber.
The team optimized the proposed microrobot-assisted wet transfer method to engineer a variety of devices. They formed electronic devices by transferring fibers with patterns onto a printed circuit board and realized electrical connections using 25 µm Au wires. They formed such electrochemical sensors for potential applications in microcatheters and other implantable devices. The scientists also demonstrated the fabrication technique using several layers of different materials to form a two-terminal graphene device on a 200 µm diameter glass fiber. They chose graphene due to the unique electrode properties of the material. They used the method to integrate active electromechanical devices onto 3-D substrates, followed by patterning on complex 3-D structures. For example, they transferred an Au pattern on a 170 µm glass capillary fiber tip microtool (engineered using two-photon lithography). The work will pave the way to engineer complex electromechanical devices at the microscale using hybrid microfabrication techniques with broad applications during precise clinical biomarker detection and precision surgery.
In this way, Antoine Barbot and a team of interdisciplinary researchers presented a practical solution to wet transfer thin films and 2-D crystals with micrometer-level precision onto 3-D substrates. They developed a floating magnetic microrobot capable of storing a preferred magnetization direction to manipulate them under a permanent magnetic field. The research team fine-tuned the robotic microgripper by adjusting the magnet’s position to grasp, align and release floating 2-D patterns.
The proposed setup is simple, reproducible and reliable. The new fabrication technique will open new applications in interventional surgery with electrochemical sensors, microactuators and complex 2-D electronic devices. The future focus for Barbot et al. will include engineering hierarchical devices based on multilayered van der Waals 2-D crystals to integrate active components onto fiber tips and ultimately form actuating and sensing capability-driven fiberbots at the nanoscale or microscale.
