Argonne chemists have identified a new catalyst that maximizes the effectiveness of platinum. Platinum—which offers unrivaled activity and stability for electrochemical reactions, such as the conversion of hydrogen and oxygen into water and electricity in fuel cells—is both scarce and expensive. Scientists are searching for alternative practical fuel cell catalysts that use far less of the costly precious metal.
In new research from the US Department of Energy’s (DOE) Argonne National Laboratory, published in Science, scientists have identified a new catalyst that uses only about a quarter as much platinum as current technology by maximizing the effectiveness of the available platinum.
Achieving high catalytic performance with the lowest amount of platinum is critical in fuel cell cost reduction. We describe a method of preparing highly active yet stable electrocatalysts containing ultralow Pt content using Co or Co/Zn zeolitic imidazolate frameworks as precursors. Synergistic catalysis between strained Pt-Co core-shell nanoparticles over a platinum-group-metal-free (PGM-free) catalytic substrate led to excellent fuel cell performance under 1 atmosphere of O2 or air at both high voltage and high current domains. Two catalysts achieved the oxygen reduction reaction (ORR) mass activities of 1.08 A mgPt−1/1.77 A mgPt−1 and retained 64%/15% of initial values after 30,000 voltage cycles in fuel cell.
—Chong et al.
In a fuel cell, platinum is used two ways: to convert hydrogen into protons and electrons, and to break oxygen bonds and eventually form water. The latter reaction, the oxygen reduction reaction (ORR), requires an especially large quantity of platinum, and scientists have been looking for a way to reduce the platinum content in oxygen reduction catalysts.
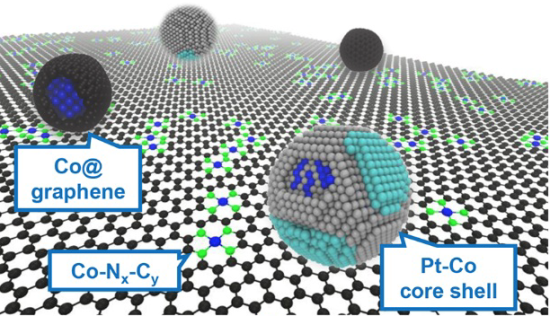
Argonne scientists found novel ways to improve platinum utilization substantially. First, they tweaked the shape of the platinum to maximize its availability and reactivity in the catalyst. In this configuration, a few layers of pure platinum atoms cover a cobalt-platinum alloy nanoparticle core to form a core-shell structure.
The core-shell nanoparticles on their own still could not handle a large influx of oxygen when the fuel cell needs to crank up the electric current. To increase the efficiency of the catalyst, the researchers relied on another approach they knew from their past work—producing a catalytically active, platinum group metal-free (PGM-free) substrate as the support for the cobalt-platinum alloy nanoparticles.
Using metal-organic frameworks as precursors, Argonne chemist Di-Jia Liu, the corresponding author of the study, and his colleagues were able to prepare a cobalt–nitrogen–carbon composite substrate in which the catalytically active centers are uniformly distributed near to the platinum-cobalt particles. Such active centers are capable of breaking the oxygen bonds by themselves and work synergistically with platinum.
As it turned out, the new combined catalyst not only improved activity but also the durability as compared to either component alone.
Liu and his colleagues have created a patented process that involves first heating up cobalt-containing metal-organic frameworks. As the temperature increases, some of the cobalt atoms interact with organics to form a PGM-free substrate while others are reduced to well-dispersed small metal clusters throughout the substrate. After the addition of platinum followed by annealing, platinum-cobalt core-shell particles are formed and surrounded by PGM-free active sites.
While the ultimate goal is to eliminate platinum from hydrogen fuel cell catalysts entirely, Liu said that the current research opens up a new direction in addressing both fuel cell catalyst activity and durability in a cost-effective way.
Since the new catalysts require only an ultralow amount of platinum, similar to that used in existing automobile catalytic converters, it could help to ease the transition from conventional internal combustion engines to fuel cell vehicles without disrupting the platinum supply chain and market.
—Di-Jia Liu
The study included computational modeling and advanced structural characterization done in part at Argonne’s Advanced Photon Source and Center for Nanoscale Materials, both DOE Office of Science User Facilities.
The research was funded by DOE’s Office of Energy Efficiency and Renewable Energy (Fuel Cell Technologies Office).
